 Limitations of Atomic Theory by JJ Thomson JJ Thomson put the great work to other scientists so that they can work on further researches using this concept. These properties of electrons are really useful to perform further studies about electrons and atomic theory in general. Electrons are able to make fluorescence when they strike objects like minerals, crystals and salts.Electrons move in the high level of velocity.Electrons can deflect if the electric filed and magnetic field are present. 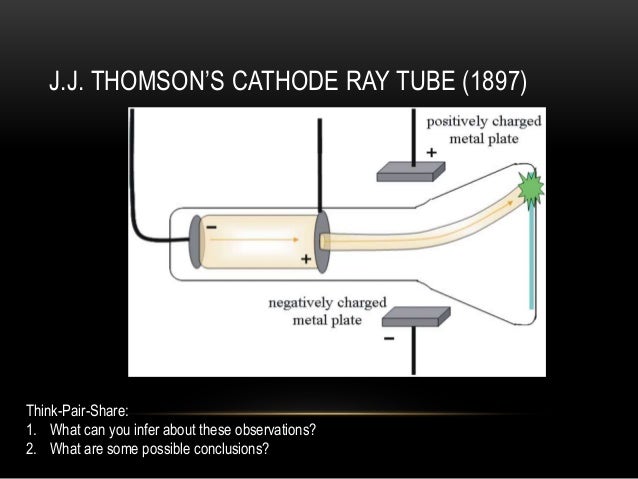
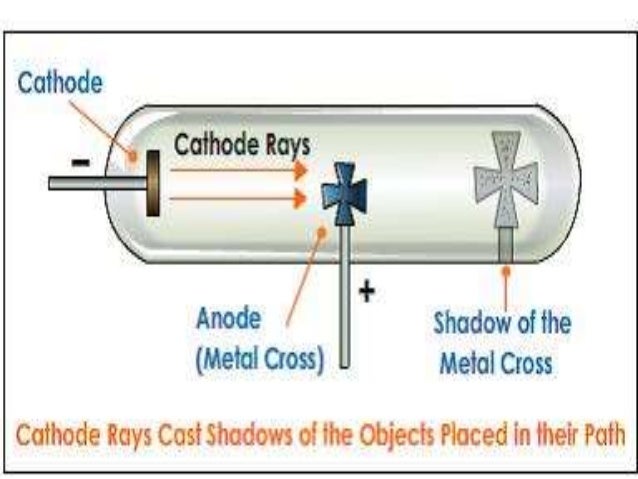
It shows that electrons can produce heat when it strikes another object. Then, when the cathode rays struck this platinum, the platinum strip will change its color to red hot. This platinum strip has the black / dark color. In the center of cathode with concave shape, you can place the platinum strip. This showed that the cathode rays or electrons have momentum and kinetic energy. Then, you will see that the wheel rotates to the anode. In the cathode rays path, you can place the light wheel of mica. Electrons have kinetic energy and momentum.So, it showed that electrons moved in the straight direction. It showed that the shadow was disappeared as the cross was placed lower. Then, the maltase cross was placed in the lower position. When heput the maltese cross on the electrons’ path, the cross’s shadow can be seen on the wall of the tube which was on the opposite side of the cathode. 
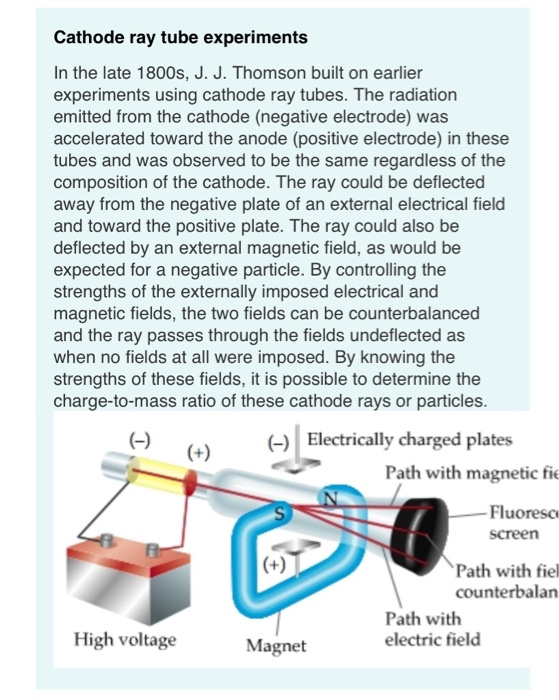
JJ Thomson conducted this experiment using the maltese cross.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
